The small heat shock proteins (sHSPs) are a vertually ubiquitous class of molecular chaperones that act in a highly efficient manner to prevent irreversible aggregation of many different protein substrates. The sHSPs are critical components of the cellular “protein quality control network”, a network of molecular chaperones and proteases that work to prevent accumulation of aberrant or damaged proteins in all cells. Studies of sHSPs are relevant to many aspects of human health because defects in protein quality control often lead to diseased states, and reduced protein quality control is associated with aging. Defining the mechanism of sHSP chaperone action, therefore, has wide-ranging implications for understanding cellular stress and disease processes.
Higher plants have 11 families of sHSPs conserved in both dicots and monocots. These include not only cytosolicnuclear proteins, but also proteins targeted to the chloroplast, mitochondria, ER, peroxisomes and nucleus. These organelle-targeted sHSPs are unique to plants. We have studied the structure and function of sHSPs belonging to the Class I and Class II groups of cytosolic sHSPs in plants using biochemical and biophysical techniques. The sHSPs are unique chaperones in that they are ATP-independent and can bind up to an equal weight of non-native protein, keeping it accessible to other components of the protein quality control network for further processing. This activity places them in the position of “first responders” to cell stress, capable of immediately binding unfolding proteins. Understanding in detail how sHSPs accomplish this feat is critical to defining their roles in protection of cells from stress. Although how these sHSPs may protect plants from heat and other stresses remains unknown, the model below presents our current thinking about how they may work. We are currently investigating the details of substrate interactions using mass spectrometry and looking at possible in vivo substrates.
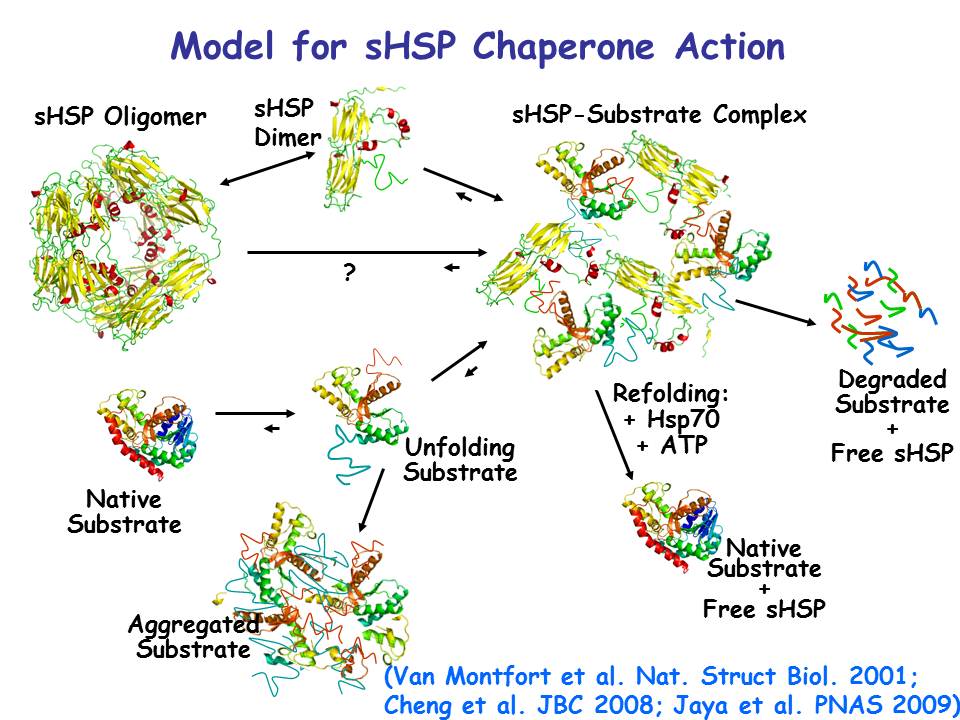
Heat-denatured proteins can either aggregate to form insoluble complexes, or can be prevented from aggregating by binding to sHsps. The denatured protein can then be degraded by proteases in the cell, or refolded by ATP-dependent chaperones like HSP70. The sHSP structure shown here is a cytosolic class I sHSP from wheat (1GME) and the model substrate is malate dehydrogenase (1MDL).